Nature Reviews Clinical Oncology: lymfoproliferatieve ziekten na CAR T-celtherapie in beeld gebracht
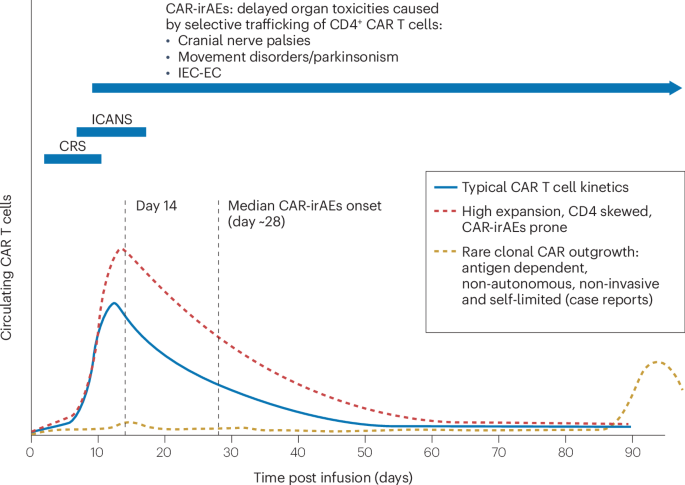
Post-CAR T-cel lymfoproliferatieve en lymfomateuze aandoeningen zijn zeldzame maar klinisch relevante complicaties met een breed en nog incompleet gekarakteriseerd spectrum. De review synthetiseert de pathobiologie, mechanistische inzichten en diagnostische uitdagingen — inclusief de complexe rol van clonale hematopoïese, infecties, immuunsuppressie en virale reactivatie.
Relevant nu CAR T-therapie breder wordt ingezet bij hematologische en solide maligniteiten en auto-immuunziekten.
Abstract (original)
Chimeric antigen receptor (CAR) T cell therapy is now widely used for the treatment of various haematological malignancies, with emerging applications in solid tumours and autoimmune diseases. Alongside its demonstrated clinical activity, this therapeutic modality has a toxicity profile that differs from those associated with traditional cytotoxic therapies, other immunotherapies, and even other cell therapy approaches such as allogeneic haematopoietic stem cell transplantation. One increasingly recognized yet poorly understood complication is the development of post-CAR T cell therapy lymphoproliferative and lymphomatous disorders, which have a clinical and biological spectrum that remains incompletely characterized. These rare events include both CAR-transgene-positive and transgene-negative lymphomas with variable and sometimes overlapping clinical features. Causal attribution is difficult, given that these proliferations often emerge in the context of clonal haematopoiesis, inflammatory or infectious triggers, immune suppression and/or viral reactivation. In this Review, we synthesize the growing body of evidence on post-CAR T cell therapy lymphoproliferative disorders, drawing on the limited but increasing number of well-characterized cases. We outline the spectrum of lymphoproliferations described so far, highlight recurrent pathological and molecular features, and discuss factors that might promote clonal expansion or transformation, including pre-existing clonal haematopoiesis, dysregulated signalling pathways, inflammatory stimuli and, rarely, CAR-transgene vector integration. A clearer framework for these disorders might improve early recognition, guide diagnostic evaluation, support treatment decision-making, facilitate classification and consensus-building efforts, and inform future mechanistic studies and pharmacovigilance efforts.
Dit artikel is een samenvatting van een publicatie in Nature reviews. Clinical oncology. Voor het volledige artikel, alle details en referenties verwijzen wij u naar de oorspronkelijke bron.
Lees het volledige artikelDOI: 10.1038/s41571-026-01147-w